Discover and read the best of Twitter Threads about #GU21
Most recents (6)
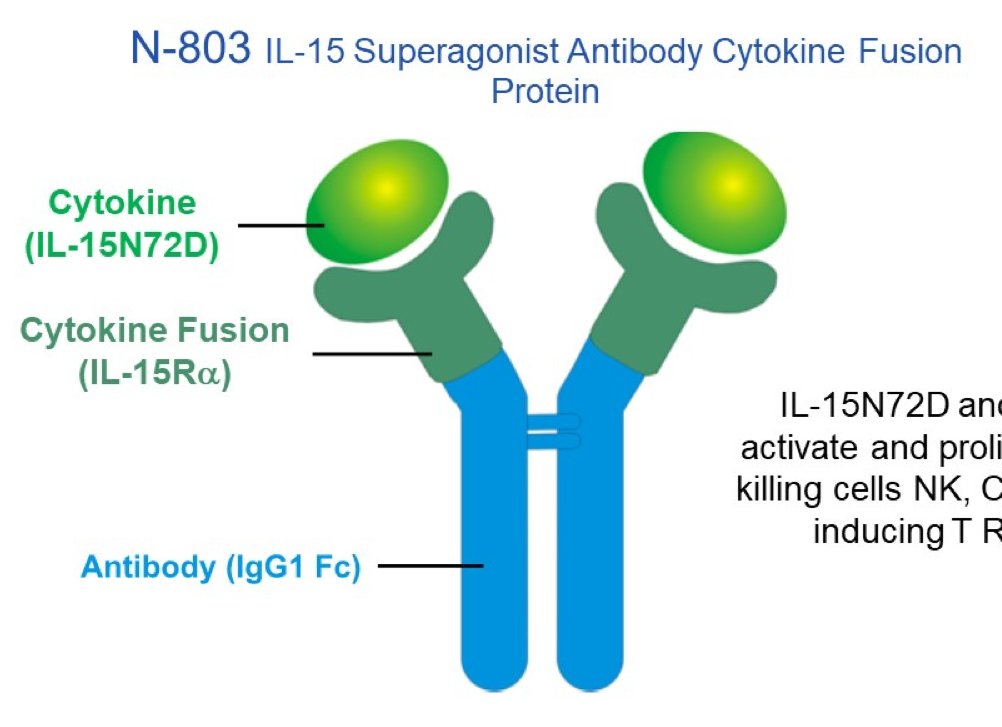
Still digesting #bladdercancer trials at #GU21. QUILT 3.032 trial clinicaltrials.gov/ct2/show/NCT03… testing #N803 in BCG-unresponsive CIS was an important result. Safety data appropriate for intravesical therapy – and CR rate was higher than seen with other drugs in similar trials.
1/n
1/n
Note, in case you missed it (as I think most of us did) that the detailed trial results were previously reported as a press release in Dec 2020 yhoo.it/3hfCcWg (unusual for a trial under ASCO embargo?)
2/n
2/n
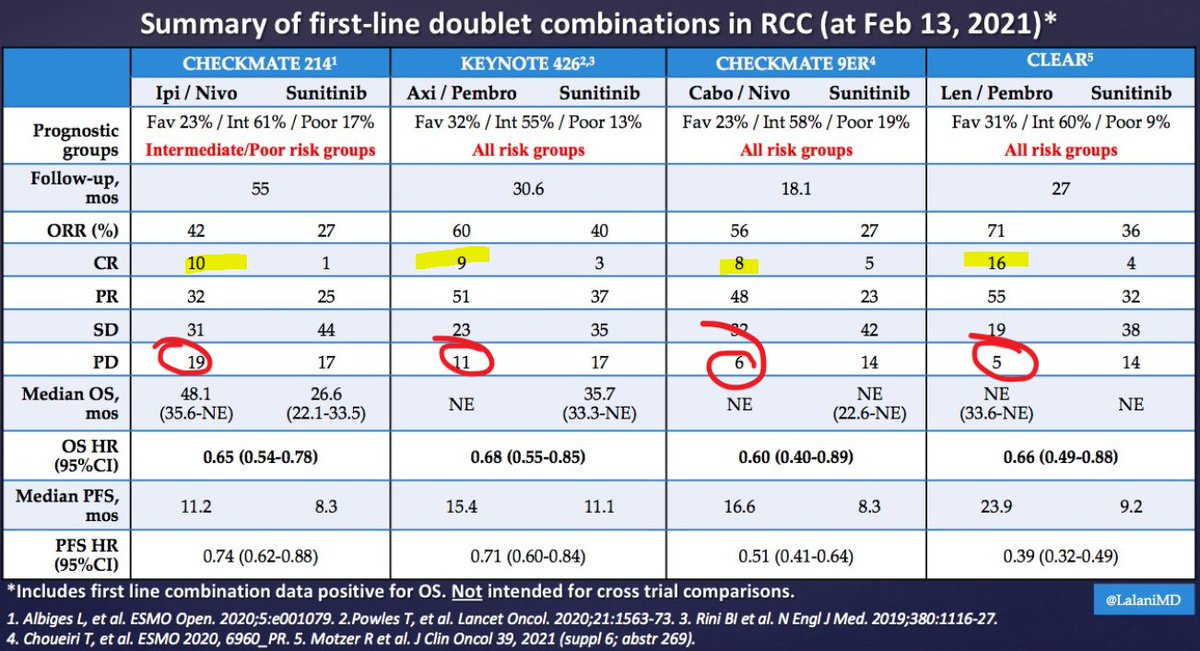
It's Monday AM post-@ASCO #GU21 & clinic starts in a couple of hours! Lots to process - I'll try to tackle optimal 1L tx for #kidneycancer. I'll make a case for cabo/nivo, leaning on the beautiful (& timely) tables below from @lalaniMD, @SoaresAndrey & @brian_rini (1/15) 


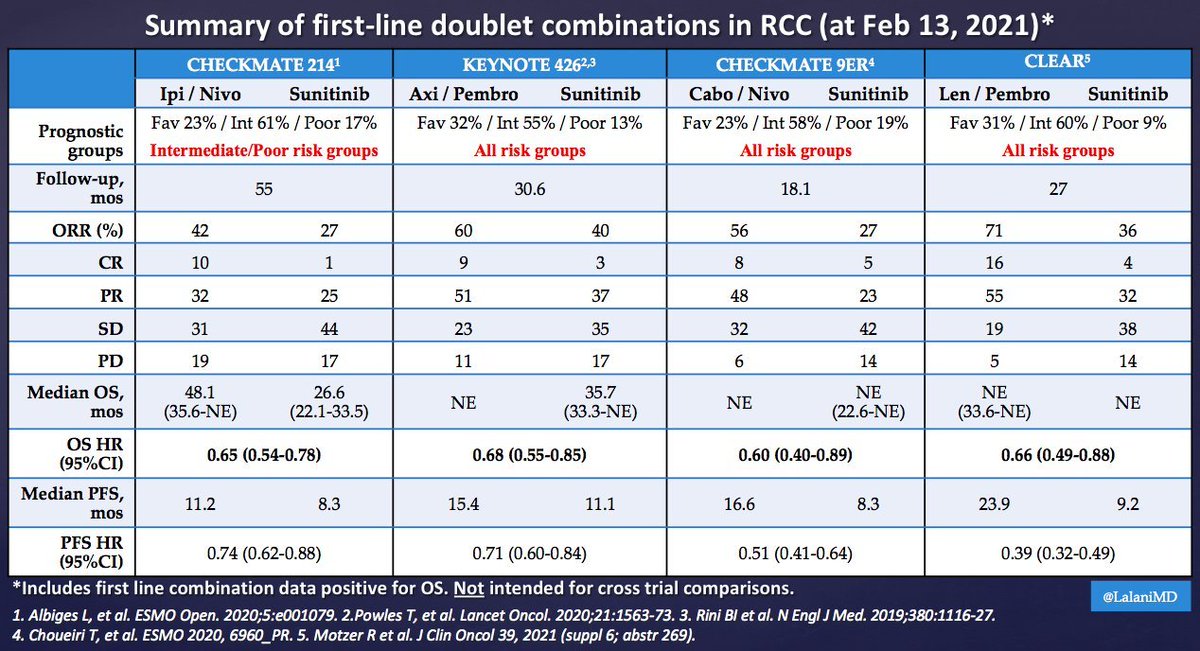

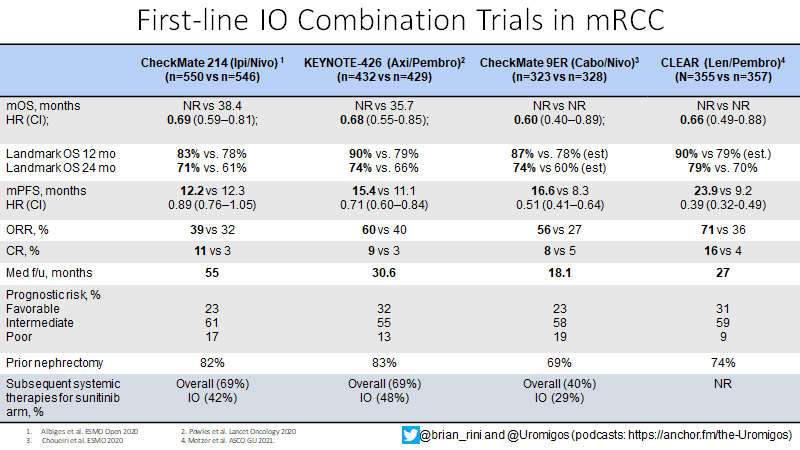
What about IO/IO? We have long f/u w #CM214 data w nivo/ipi, no doubt (@AlbigesL et al in @myESMO Open). And treatment-free interval discussed by McDermott @BIDMChealth is no doubt impt. But we've known data not as impressive for favorable risk (2/15) 



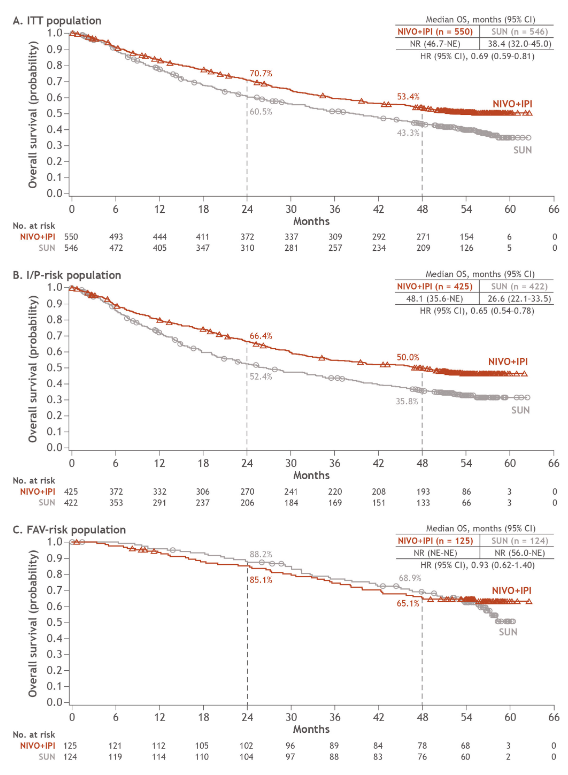

And furthermore, as @ERPlimackMD points out in another tweet, impt to look at primary PD rates (seen in @lalaniMD's table) - nivo/ipi at 19%!!! CR rate used to be something we highlighted w nivo/ipi, but now comparable across studies (3/15) 
What a fantastic way to end #GU21! A year ago, I attended my 1st #ASCOGU. A year on, I am so grateful to all pts, #IMDConline collaborators, @ASCO, & @DrDanielHeng for the opportunity to present the outcomes of 1L IO Combos in mRCC.
@OncoAlert @DrChoueiri @ShaanDudani @montypal



@OncoAlert @DrChoueiri @ShaanDudani @montypal




Excited to share the results of @SWOG 1500, which we are presenting at @ASCO #GU21 & reporting in @TheLancet today. 1st, THANK YOU to patients & families. 2nd, hats off to a tremendous group of investigators. This was a team effort! Wanted to share how it all began: (1/13) 





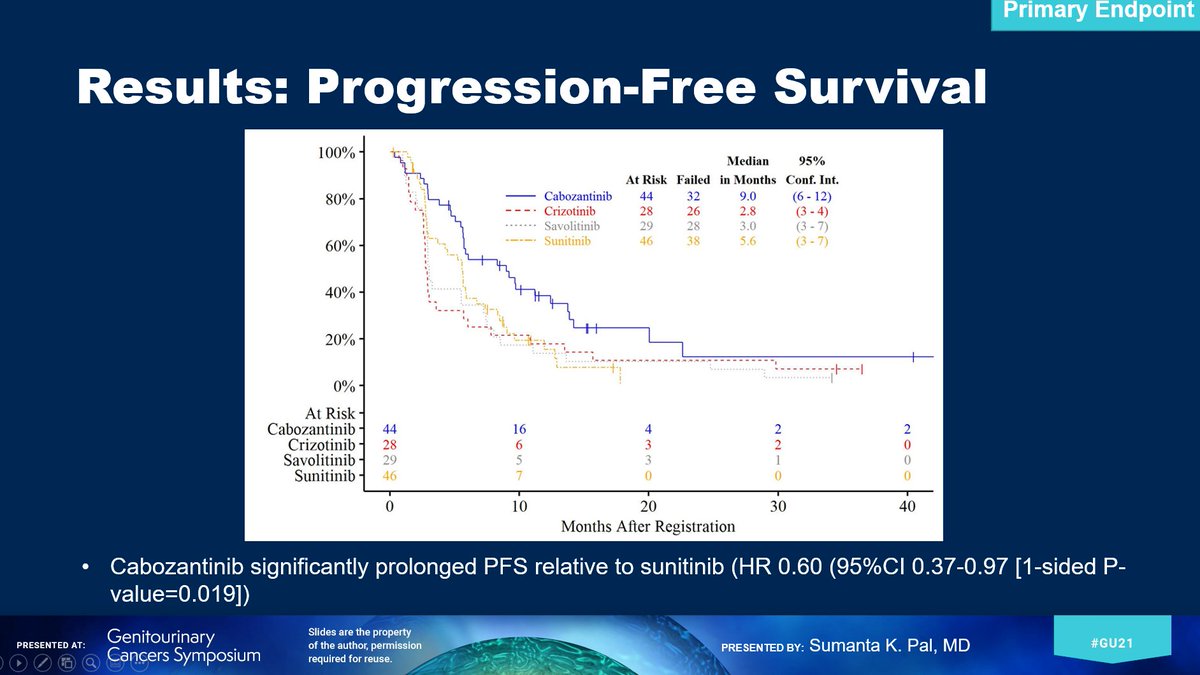

A telling snapshot from my "S1500 folder" on my computer. With mentorship from @PrimoLaraMD I submitted the concept 1st in 2012. Had advice from my mentor @DrChoueiri at the time as I conceived of the study design - interestingly, the proposal was XL184 (cabo) v sunitinib! (2/13) 

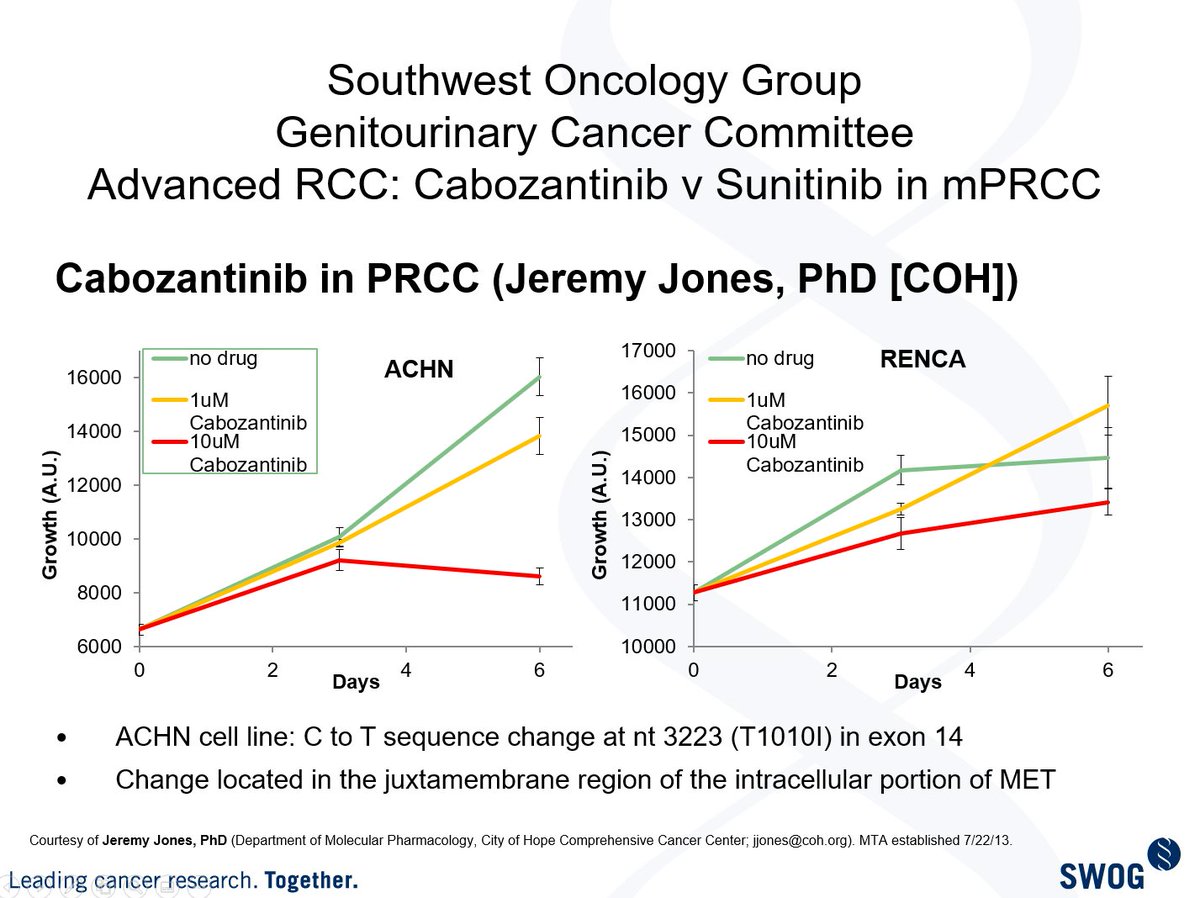
My dear friend @Jeremy_O_Jones helped me w some preclin work & with emerging evidence from the P1 trial that @DrChoueiri #DrMcDermott #DrJanDutcher & I (along with others), we proposed the following formal design to the group in 2013: (3/13) 




My take on #CheckMate274: Is adj CPI now SOC for #MIUC? Not yet. Here's the design presented today at @ASCO #GU21 - first, thanks to patients & investigators (#DrDeanBajorin, @MattGalsky) who should be commended for completing an adj trial in #bladdercancer - no small feat! (1/9) 

First, a trip down memory lane. Here is the data from IMvigor010 - adj atezo v observation. No diff in DFS/OS, period. No difference based on PD-L1 status. Could it be PD-1 v PD-L1? Hard to prove. Could it be obs v pbo control? (2/9) 







I stole this slide from @MattGalsky in a separate thread i came across. Here he nicely highlights the attrition on control and experimental arms. IMvigor010 lost a substantial proportion of folks due to "other" reasons on the observation arm. (3/9) 

1/6⭐️🚨Molecular Profiling of Bladder Cancer Early-Phase Clinical Trial Participants Predicts Patient Outcomes published as Rapid impact paper @oalhalabimd et al
@MCR_AACR @AACR @CancerMedMDA #GU21 @ASCO @OncoAlert
#bcsm bit.ly/3d25A1U
@MCR_AACR @AACR @CancerMedMDA #GU21 @ASCO @OncoAlert
#bcsm bit.ly/3d25A1U

2/6 Prognosis for patients with metastatic bladder carcinoma (mBC) remains limited and in need of novel therapies @OncoAlert @MCR_AACR @oalhalabimd . #bcsm bit.ly/3d25A1U
3/6 Patients' tumors or circulating tumor DNA were analyzed by NGS. The median progression-free survival was 4.2 months, the median overall survival was 9.6 months, and the overall response rate was 17.5%. #GU21 @MCR_AACR @oalhalabimd #bcsm bit.ly/3d25A1U